Plating FAQs
What is electroplating and how does it work?
Electroplating is the process of utilizing electricity as the energetic driving force to promote redox reactions in which a thin layer of metal is deposited on top of a conductive substrate.
In practice, parts to be plated are submerged in an aqueous bath with dissolved salts for increased conductivity. Direct current is applied with the parts to be plated made cathodic and the sacrificial replenishment metals made anodic.
At the anode, the sacrificial metal undergoes an oxidation half-reaction, releasing electrons and becoming positively charged metal cations. The metal cations are made aqueous soluble by complexation with appropriate ligands and are dispersed uniformly in the bath through diffusion and solution movement.
At the cathode, metal cations approaching the surface of the part to be plated accept free electrons from the electrically biased circuit and are reduced. This process deposits a layer of metal from the bath at all conductive interfaces, and in turn, plating the part.
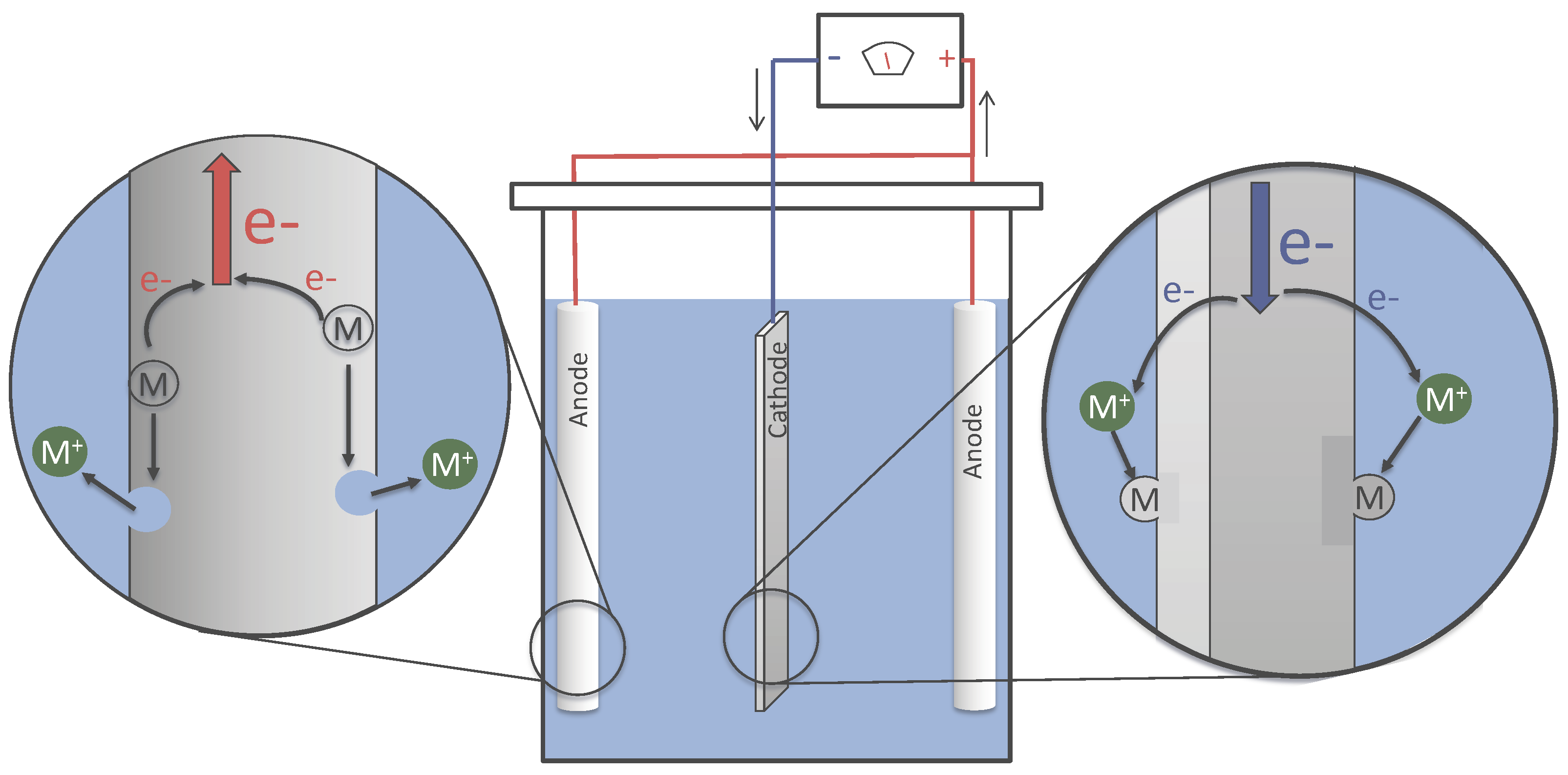
Schematic Diagram of an Electroplating Tank Showing the Flow of Electron and Metal
How does electroplating differ from “electroless” plating?
Electroplating, as the name suggests, utilizes electricity as the energetic driving force to produce a thin layer of metal or plate on a part. The thickness of the plate produced is dependent on the amount of current applied to the part as well as the time the part is in the plating bath.
Electroless plating on the other hand does not require electricity. These processes are autocatalytic in nature, meaning that the metal deposited as a plate also serves as a catalyst for further deposition of metal. Thus, this process does not depend on electricity and the thickness of the deposit is solely a function of bath chemistry and submersion time.
What is immersion plating and how does it differ from electroless plating?
Technically, immersion plating is a type of “electroless” plating in the sense that it is a plating process that does not require electrical current. However, the two types of processes differ in an important way:
The term electroless plating typically refers to an autocatalytic process whereby the metal plated catalyzes further plating. This process occurs indefinitely, and the plate gets thicker and thicker, until either the part is removed from the bath, or the bath runs out of metal.
Immersion plating differs from electroless in that it is not autocatalytic and requires exposure to the base substrate to promote the plating process through difference in redox potentials. Once full and complete coverage of the substrate is achieved, there is no longer an electrochemical driving force and the plating process subsides. Thus, immersion coatings are typically very thin. Often in the metal finishing industry immersion plating is avoided as the adhesion of the plate is subpar. However, specialty baths designed for immersion plating eliminates this drawback, providing adequate adhesion. A Square offers gold plating utilizing this technology. This bath is capable of plating pure soft gold from 4-10 microinches thick.
How uniform is the thickness of an electroplated deposit?
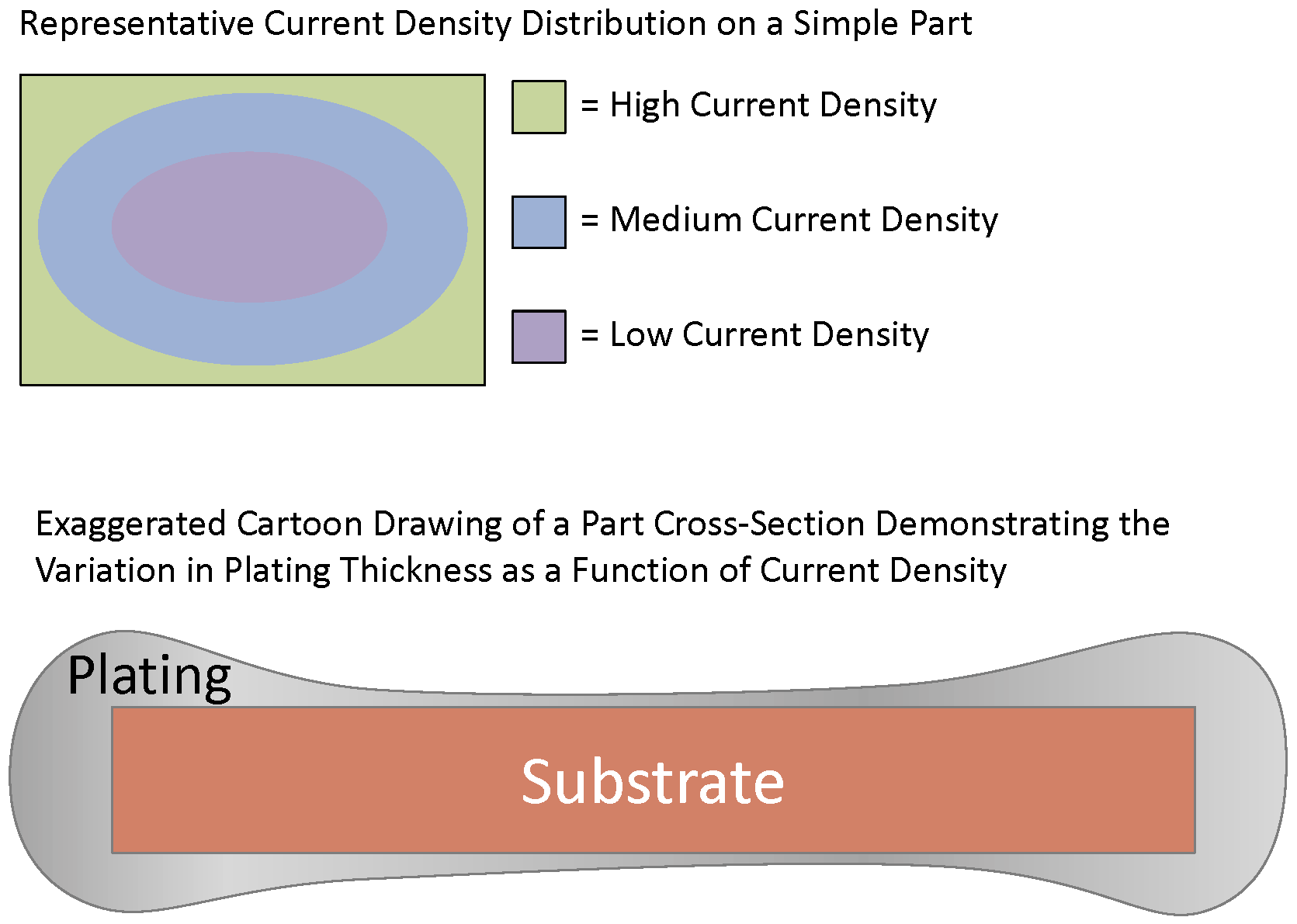
The uniformity of plate thickness is highly dependent on the design of the plating system, electrical contacts, and the shape and dimensions of the part. Electroplating is a current dependent process. Thus, the greater the current to a specific area of a part the faster the plating layer will be built up in that area. Areas of high current density, such as sharp edges, receive more plating, whereas those with low current density, such as the middle of large parts, receive less plating. Some features, such as blind holes, are particularly troublesome to achieve any substantial plating thickness and it is typical for these areas to be completely free of any plating.
As a general rule of thumb there is about a ± 50% variation between the nominal thickness and the high and low points on the part. In most applications excess plating is not an issue and engineers will choose to specify a plating minimum instead of a plating range or maximum. In instances requiring a narrow plating range, specialty racks and masks can be designed to help facilitate plating uniformity but this in turn will increase the part cost and/or NRE costs of plating.
What is A Square’s typical lead-time?
Our standard lead time is two weeks. We understand that from time to time “super hot” jobs will come along that just can’t wait. In these situations, we work directly with our customers to arrange a production schedule that satisfies their production requirements.
What is a “typical” plating thickness and specification?
We adamantly believe in customer directed plating. Every customer, part, and application is different. Thus, there is no one-size fits all solution to metal finishing. The wear demand on the part, local environment, visual aesthetics, performance requirements, material properties of the plate, and etc. all contribute to the ideal metal and thickness of the plate. Please consult with our engineering team to devise a metal finish that best suits your projects budget and performance requirements.
For reference, below is a list of standard specifications for various metals that we plate:
Silver: | Gold: | Nickel: | Tin: |
|---|---|---|---|
QQ-S-365 | MIL-G-45204 | QQ-N-290 | ASTM B545 |
ASTM B-700 | ASTM B488 | ASTM B-733 | MIL-T-10727 |
AMS 2410 |
What finishes does A Square provide?
A Square specializes in precious and semi-precious metal plating. We offer silver, gold, tin, nickel, electroless nickel, and copper plating. Please see our finishes page for more details about the finishes we offer.